What is the Crystallization of Cannabinoids?
Crystallization is a process when molecules arranged into a well-defined, rigid crystal lattice to minimize their energetic state due to the fact that a Crystal form is a very low energetic state. The most known cannabis crystal is CBD. There is a wide range of additional Cannabinoids that can be isolated and purified as part of the crystallization process- THCa, CBDa, CBDv and CBG to name a few.
The cannabinoids isolation and crystallization process begins after multi-stage separation and purification processes of the Cannabis flowers and extraction, usually using isolation techniques.
Cannabinoids Crystallization process mechanism
The “driving force” behind the Cannabinoids crystallization process is supersaturation. Supersaturation is a physical state, where Cannabinoids molecules suspended in a solvent until it exceeds the solubility limits, and as a result of this physical state, it is being precipitated out of the solvent to decrease its energy state. Surely- there are few cannabinoids that their crystallization process is much more complicated due to their molecular structure and physical properties. These molecules tend to “oil out’, and create a solvent phase separation in supersaturation conditions.
What can be manufactured using Cannabinoids isolates?
Crystallization is an essential process in the Cannabis and Hemp industries. It purifies the active material compounds (cannabinoids), concentrates it, increasing its stability and reduces it’s particle size distribution and volume, and achieving >99% purity level.
A “good” Crystallization process should eliminate impurities, have a high yield and be robust and scalable. Cannabinoids isolates/crystals comply with the GMP (Good Manufacturing Practice) standards and can be used even as an API in the pharmaceutical and industries.
Crystallization process types
There are several cannabinoids crystallization techniques that can be used in order to produce Cannabinoids isolates. The preferred technology can be chosen based on several criteria such as technical barriers, capacity, space, target market, regulatory requirements, purity level, yield and process development capabilities.
The crystallization process in a multi-stage process and actually being divided into two main stages.
Cannabinoids crystallization technology- The first stage
We can define two main types of crystallization processes for the first crystallization stage, based on the crystallization process engineering: “Static Crystallization” and “Stirred Crystallization”.
Static crystallization of Cannabinoids
In the static crystallization technique, Cannabinoids crystals formed in a non-kinetic process under cooling conditions. This process is more likely to be used for small-scale isolation processes. Before crystallization cooling stage initiation, we should heat the solvent to create supersaturation of the Cannabinoids. Afterward, we cool the supersaturated solution to a specific temperature and cooling rate to produce “Diamonds” which later on, being carved from the crystallization dish. This type of crystallization usually requires rigorous further processing to reach the required particle size.
Stirred crystallization of Cannabinoids
The Stirred crystallization technique, on the other hand, is in kinetic terms, a quicker process due to additional physical forces that accelerate the crystallization process rate. Moreover, the cannabinoids stirred crystallization process is more scalable, and in case performed properly, can reduce time and additional processing and purification stages.
Cannabinoids crystallization technology- The second stage
There are four types of crystallization options that can be adopted after the first stage of crystallization process completion:
- Cooled Crystallization
- Anti-solvent Crystallization
- Evaporative Crystallization
- Co-crystal crystallization.
Cooled Crystallization of Cannabinoids
The Cooling crystallization technology is more relevant for Cannabinoids molecules which considered as “not heat sensitive”, and as a result, these molecules will be soluble at high concentrations at high temperatures, and will decrease in solubility dramatically at low temperature required for the crystallization process. The cooling crystallization is relevant for cannabinoids such as CBD.
Anti-solvent Crystallization of Cannabinoids
The Anti-solvent crystallization process is relevant for Cannabinoids that have good solubility characteristics at low temperatures. In the Anti-solvent crystallization process, we will usually use two different solvents – the first solvent will solubilize the cannabinoid molecules, and through graduated addition of the second solvent, we will create supersaturation that will create cannabinoids precipitation and crystallization.
Evaporative Crystallization of Cannabinoids
The evaporative crystallization technique is relevant for cases where we deal with Cannabinoids that have high solubility in low temperatures, and we have no anti-solvent that can be used. In evaporative crystallization, we will gradually remove the solvent through evaporation to create supersaturation and cannabinoids crystals.
Tips for the cannabinoids crystallization process
- Process development- When it comes to the proper crystallization process, there is a need to define clearly what will be performed exactly at each stage and what are the critical parameters to be kept throughout the process.
- Control- A proper crystallization process engineering should be controlled properly. Poor process control, especially over critical process parameters will lead to low yields, low purity and non-GMP (Good Manufacturing Practice) product that also will require additional processing steps.
How to begin with cannabinoids crystallization process development?
- Always make sure to work in a fume hood and use protective equipment
- Solvent choosing – Solvent choosing should be based on its toxicity, environmental impact, ease of evaporation and cost.
- Solvents screening- Take a pea size of purified cannabinoid extract and place it in a test tube. Then add about 3 ml of the chosen solvent and shake vigorously. In case the sample dissolves in-room temperature, this is not a good solvent to continue to work with.
- Start heating the tube and watch what happens. In case molecules start to dissolve as you reach a higher temperature, the solvent might be appropriate for Crystallization process.
- Let the solvent cool down to room temperature and inspect visually to verify crystallization occurred.
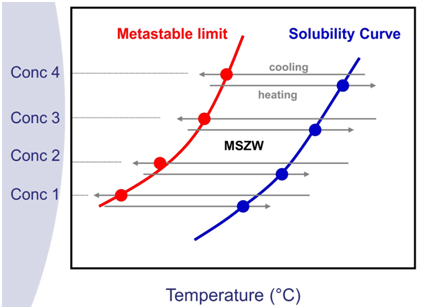
- Creating a solubility curve – Now you will need to check the correlation between the solvent temperature and the solubility. The solubility of a molecule in a solvent is a thermodynamic parameter, and it is the most important factor in the Crystallization cooling process design.
Source :
https://nikyang.com/en/automated-solution/parallel-crystallizers-for-drug-development/
- The solubility curve can be established through the process of preparing known concentrations of the chosen Cannnnabinoid in the solvents we have screened and measure the temperature in which it dissolved entirely.
- Cool the solutions to create crystallization- The metastable limit is “the zone” between the solubility curve and crystallization point is called “metastable zone width” or shortly MTZW. In other words, this is the range where our Crystallization process will take place. CBD for example, will be more soluble in Pentane at higher temperatures and will demonstrate limited solubility characteristics at lower temperatures, causing crystallization.
Next steps:
- Create cooling gradient, seeding strategy, filtration and drying to remove solvents residues.
- Test the Cannabinoids crystals in a certified laboratory.